In Search of a Cure for Low Enrollment in Oncology Clinical Trials
Blog
Patient Engagement & Recruitment


Patient education is key in overcoming barriers to participation in oncology clinical trials.
Clinical trial recruitment and enrollment remains challenging across therapeutic areas, and this is particularly the case in oncology. According to data obtained from accreditation information submitted by the Commission on Cancer programs in the US, enrollment in cancer clinical trials was 7%,1 slightly higher than historical estimates of less than 5%. A meta-analysis of oncology studies conducted in the US revealed that in 56% of cases low enrollment was due to lack of available trials, 22% of patients did not meet eligibility criteria, and 15% of those who were eligible did not enroll.2
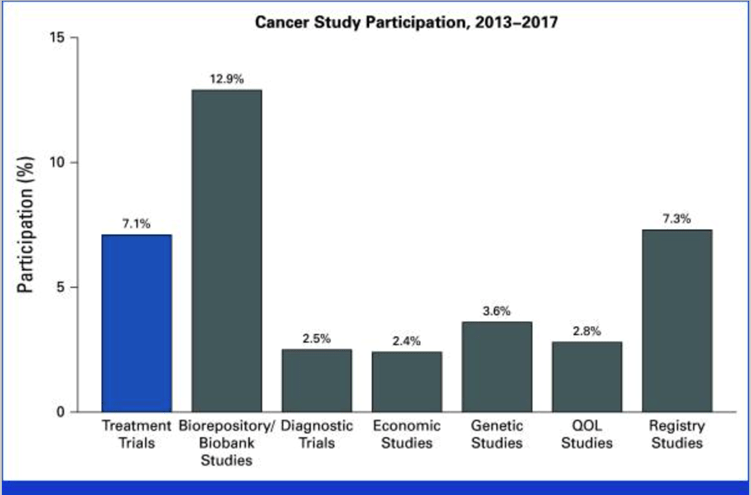
Estimated cancer study participation in the US by type of study, 2013-2017.
The estimate for treatment trial enrollment is highlighted in blue. QOL, quality-of-life.
Source: Journal of Clinical Oncology1
For global oncology trials, the participation rate is even lower. Despite exponentially increased industry investment in oncology R&D, patient enrollment in clinical trials remains below 5% globally.3
Lower enrollment brings with it a hefty price tag. Ross Pettit, Chief Development Officer of Kestrel Therapeutics, estimates that the average cost of delayed recruitment in an oncology clinical trial is $175,000 to $250,000 per patient.
Prior treatment can be a determining factor in oncology clinical trial participation. Eligibility guardrails are designed to ensure both scientific validity and patient safety, the latter due to drug tolerance and tumor biology. Patients who have received too many prior therapies often fall into the “exclusion” category. Trial protocols usually require a "washout" interval of about four weeks between the last dose of a prior therapy and the start of a new trial.
Friends of Cancer Research advocates for broader, more inclusive eligibility criteria to improve patient access and reduce health disparities. The organization worked with the American Society of Clinical Oncology (ASCO) to develop recommendations for broadening eligibility criteria, leading to development of US Food and Drug Administration (FDA) guidance documents on how eligible patient populations are defined.
Will Franco, Citeline Senior Solution Consultant, notes that artificial intelligence plays a key role, especially in rare disease and in oncology clinical trials, because “we need to find a treatment fairly fast for these patients. If we don't identify these patients quickly, we’ve missed the window of opportunity for them to participate in the trial.
“We don't hear how challenging it is for someone to participate in the trial even if they want to participate. A lot of this is due to those timelines.”
Rural patients in particular are significantly underrepresented in cancer clinical trials. A systematic review of the literature4 revealed that barriers to participation by rural populations are present at the patient, physician, and healthcare system levels.
Notable barriers — common among oncology clinical trials in general as well as other therapeutic areas — fall into three categories:
Compounding the overall problem of patient enrollment in oncology trials is low participation among minority groups. The American Association for Cancer Research 2024 Disparities Progress Report revealed that Black, Hispanic, Native American, and Asian patients were routinely underrepresented:

In addition to sponsor websites and government registries such as ClinicalTrials.gov, patients often turn to advocacy groups for education and support. These groups provide disease-specific resources for patients in search of answers. The National Comprehensive Cancer Network is one place patients can search for support groups by disease.

Amanda Hollinger
Amanda Hollinger, Executive Director of the Head & Neck Cancer Alliance, a patient advocacy organization, says improving health literacy around clinical trials and reducing the stigma and fear around them — “those are all things that play a role in bridging the gap between patients and the research pipeline.
“I still think there are existing preconceptions or misunderstandings about what clinical trials are. We try to provide education about what they are and they are not, so that patients are aware that if they're in a clinical trial, they will still receive the standard of care. It's not that they're not going to be treated or that they're a guinea pig or anything like that.”
The alliance regularly offers educational webinars for patients, in addition to other educational content and a clinical trial finder. Other patient resources include peer-to-peer mentoring support, an online support community with over 13,000 members, limited financial assistance, and an annual survivorship symposium. Patients also can fill out an online form to request a contact or call a toll-free number to speak with a patient resource navigator. While her organization provides a wide array of resources, Hollinger says, “I think patients are far more likely to join a trial when a trusted healthcare provider tells them about it and encourages them to consider it.”
She says restrictive inclusion/exclusion criteria and fear of getting a placebo are two reasons for low trial enrollment. She notes that patients often have trouble finding relevant trials, perhaps because they are using the wrong search terms. That’s what happened to Bess Stillman, an ER physician, who struggled to find a clinical trial for her husband, who was diagnosed with esophageal cancer.
Hollinger adds that head and neck cancers present their own set of obstacles for patient participation, such as certain comorbidities or reduced quality of life. “There are often significant treatment-related impairments,” she adds, “everything from difficulty eating, speaking, swallowing, weight loss, weakness — things that can make travel or participation burdensome.”
She emphasizes communicating the benefits of participation to patients. Clinical trials “really are opportunities to access tomorrow’s medicines today, as well as … help other people down the line.”
Why are oncology clinical trial enrollment rates so low?
Cancer patients are slow to enroll in clinical trials due a lack of available trials, strict eligibility criteria, fear of participation, medical mistrust, lack of awareness, and geographic challenges, among others.
How does prior treatment affect a patient's eligibility for a clinical trial?
Patients who have undergone too many prior therapies are often excluded from trials due to concerns about drug tolerance and tumor biology. Most protocols also require a "washout" period of approximately four weeks between the last prior therapy and the start of a new trial, which can further limit who qualifies.
What is the financial impact of slow clinical trial recruitment in oncology?
Delayed recruitment carries a significant cost. Delayed recruitment in an oncology clinical trial is estimated to cost $175,000 to $250,000 per patient.



1Unger JM, Shulman LN, Facktor MA, Nelson H, Fleury ME. National Estimates of the Participation of Patients With Cancer in Clinical Research Studies Based on Commission on Cancer Accreditation Data. J Clin Oncol. June 20, 2024. Available from https://www.citeline.com/en/resources/2026-pharma-predictions [Accessed April 30, 2026]
2Systematic review and meta-analysis of the magnitude of structural, clinical, and physician and patient barriers to cancer clinical trial participation. Unger JM, Vaidya R, Hershman DL, Minasian LM, Fleury ME. J Natl Cancer Inst. 2019. Available from https://pmc.ncbi.nlm.nih.gov/articles/PMC6410951/. [Accessed April 30, 2026]
3Bloomberg New Economy International Cancer Coalition, McKinsey Cancer Center, Cure4Cancer; Advancing Global Health Equity in Oncology Clinical Trial Access. Cancer Discovery. Dec. 1, 2024. Available from https://aacrjournals.org/cancerdiscovery/article/14/12/2317/750143/Advancing-Global-Health-Equity-in-Oncology. [Accessed April 30, 2026]
4Thapar R, Mulley R, Ferriola N, Luan T, Sanker R, Wong L, Abel EL, Go L. Barriers and facilitators to recruitment of rural patients to cancer clinical trials. Proc (Bayl Univ Med Cent). Aug.25, 2025. Available from https://pubmed.ncbi.nlm.nih.gov/41341082/. [Accessed April 30, 2026]

To enable the booking feature, please enable all cookies in your browser.