I Survived the SCOPE Summit … and Have Key Takeaways to Prove It
Article
Clinical


Another SCOPE Summit is in the record books, and it was jam-packed with informative sessions. Themes included patient-centricity, AI (no surprise here), patient recruitment, and much more. See our take — and takeaways — on this annual industry event.
If you’ve never been to the annual SCOPE Summit, it can be intimidating and overwhelming. This year’s summit marks my third, so I take it all in stride. Amidst the sea of faces and rows upon rows of exhibitors, I was able to recognize — and reconnect with — many pharma folks. While I’m still recuperating (both physically and mentally), I’d like to share a few highlights while they are fresh in my mind. Let’s do this!
Patient-centricity is no longer just a buzzword; it’s being put into practice at all stages of the R&D process. Just ask Jennifer Miller, on staff at the Yale School of Medicine and CEO of the Good Pharma Scorecard, an index that ranks pharmaceutical companies on their bioethical performance. She also is founder of the nonprofit Bioethics International.
Miller’s scorecard ranks pharma companies on transparency, data sharing, access to medications, and diversity, equity, and inclusion (DEI). This year, a fifth scorecard is being added: patient-centricity.
Data are still often siloed, fragmented, and dispersed. This theme was echoed in the presentation “Why Source Matters,” led by Shawn Murphy, Chief Research Information Officer at Mass General Brigham.
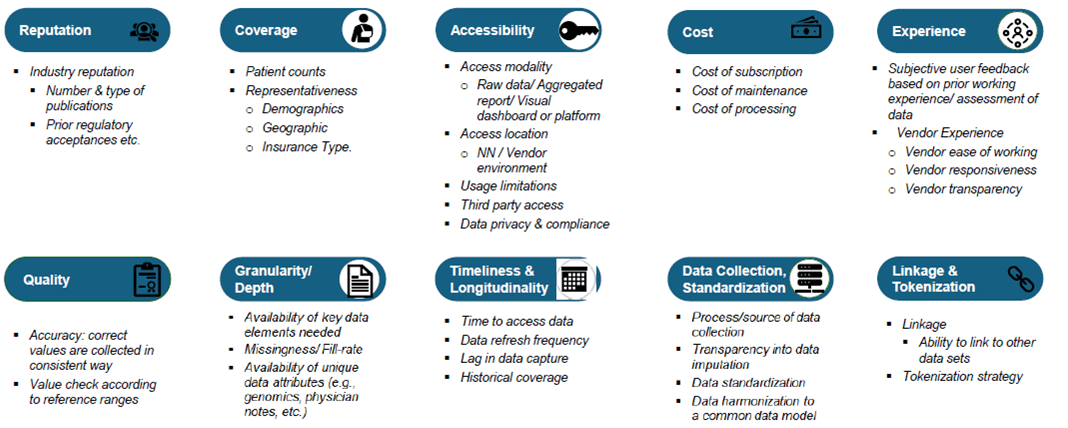
In the session “Fit-for-Purpose RWD,” Thomas Dougherty, Real-World Data Strategy Lead at Novo Nordisk, emphasized the importance of selecting a data vendor. He outlined several categories to consider when evaluating data fitness of vendors:
What industry insiders already know, patient recruitment is a major a roadblock for most clinical trial sponsors. In a keynote panel discussion — “From Years to Months: Is Radical Acceleration in Clinical Research Possible?” — Jared Saul of Amazon Web Services (AWS) lamented, “We don’t know where the patients are.”
If you ask any of my colleagues, they would heartily disagree with that statement. With our Citeline PatientMatch solution, we alert CROs, investigators, or site staff when a patient precisely fitting the protocol is present at their site.
However, Saul noted that it is possible to find patients without exposing their personally identifiable information (PII). My colleagues and I totally concur on that point, thanks to data anonymization best practices.
At Citeline, feasibility and patient recruitment go hand in hand, as reflected in two of our sessions, “Design with Enrollment in Mind: The Feasibility-Informed Protocol” and “Start with Patients, End with Confidence: Rewiring Feasibility for Real‑World Enrollment.” In the first session, Claire Riches, Citeline VP of Clinical Solutions, echoed the Citeline mantra of “plan, find, enroll,” reiterating the importance of a holistic approach to patient recruitment. Riches was joined by Melissa Harris, Global Head of Patient Recruitment & Engagement at Fortrea, who discussed the inherent problems with so-called unicorn protocols. “A unicorn protocol is really pretty and it sparkles,” she said, but “you can’t recruit for one of those,” Harris said.
In the second session, Christina Masturzo, SVP, Clinical & Regulatory Product, and Anjani Varma, Senior Product Marketing Manager, shared a case study involving a Phase III cancer study. Using an AI-enabled protocol analysis, Citeline implemented a three-tiered analytical strategy to optimize inclusion/exclusion (I/E) criteria, validate US sites, and prepare for global expansion. Citeline’s methodology encompassed examining the competitive landscape, analyzing over 650 investigators, reviewing the organization’s trial history, and incorporating real-world patient data. The sponsor’s US sites were validated, revealing an 89% overlap between its sites and the Citeline-recommended sites. In addition, Citeline:
In the end, this sponsor gained the data it needed to back up its decisions and the confidence it needed to proceed with the trial.
AI was here, there, everywhere. Fortrea’s Harris clarified a popular caveat when working with AI: “We need a human in the lead, not in the loop.”
Suzanne Caruso, Citeline GM & Executive VP for Clinical & Strategic Intelligence, shared keen insights in the Citeline session “From Insights to Outcomes: A Practical AI Playbook for Trial Teams.” Caruso said the human-enabled clinical trial workflow “is where I think AI has the biggest opportunity to be able to help us.” She calls this the fragmentation gap between where the data and the knowledge have sat and the actual workflow.
Caruso added that other areas where pharma can benefit from AI include recommendation models, predictive models, and forecasting. “Generative AI is great for parts of protocols,” she noted.
For 2026, however, Caruso said AI agents will be the overriding theme. “The AI agents are your best friends.” These agents are what will be driving the clinical workflow, she said.
“We're seeing a lot of resonance in the natural language processing [NLP] of unstructured data.” NLP is particularly helpful for claims data and physician notes, she said.
Caruso also touched on recommendation engines, saying they can help with investigator selection, protocol prediction, eligibility criteria, endpoint recommendation, and secondary endpoint recommendation. Machine learning models support one’s recommendation engines, she explained.
AWS’s Saul said another benefit of AI is that it helps move across siloed datasets. This is extremely relevant for clinical trials; in addition to the challenges listed above, data sources are both numerous and disparate.
Justin North of TriNetX, a strategic partner of Citeline’s parent company Norstella, properly summed up the current state of AI in pharma: “Everyone’s kind of throwing AI against the wall and seeing where it sticks.”
In today’s political climate, the scientific need for clinical trial diversity is overshadowing the ethical need. Tyler Allen, Program Leader for Clinical Research Representation at Duke Cancer Institute, spoke about barriers to clinical trial participation for underrepresented groups. He said the industry must address logistical barriers including transportation, housing, and financial strain. “These are structural problems. They are not patient limitations.”
Allen also emphasized the importance of health literacy and the need for plain language in patient-facing communications.
Tobias Krause, Managing Director-Europe for SubjectWell, made the case for digital outreach — social media and search engines — in engaging underserved populations in clinical trials. He presented two case studies in which an online questionnaire was used to identify “under-the-radar” patients.
Another diversity-related session focused on site readiness. Ramona Burress, Chief Development Officer of ONYX Collective, reiterated Allen’s comment on systemic issues when speaking about sponsor-site gaps. She pointed to infrastructure. “The problem is in systems, not people.”

Darcy Grabenstein
Director of Content Strategy and Thought Leadership | Citeline
Darcy is the Director of Content Strategy and Thought Leadership for Citeline. A journalist by training, she has more than 30 years of experience in marketing, advertising, and public relations.
What were the biggest themes at the SCOPE Summit this year?
Dominant themes included patient-centricity, data quality and integration, patient recruitment challenges, the expanding role of AI, and clinical trial diversity. Across sessions, speakers emphasized moving from theory to execution, particularly in how sponsors design protocols, use real-world data, and engage patients more effectively.
How is patient-centricity being measured and applied in clinical research today?
Patient-centricity is no longer just aspirational — it’s being actively evaluated and implemented. At SCOPE, it was highlighted as a new scoring dimension in the Good Pharma Scorecard, alongside transparency, data sharing, access to medicines, and DEI.
Why does patient recruitment remain such a major challenge for clinical trials?
Recruitment continues to be a bottleneck due to overly restrictive or unrealistic protocols, limited visibility into where eligible patients are, and logistical barriers for participants. A feasibility informed, data-driven approach — one that balances protocol rigor with real-world enrollment realities — is essential.




To enable the booking feature, please enable all cookies in your browser.