The Ins and Outs of Decentralized Clinical Trial Technologies
Article
Clinical


Technology is the backbone of decentralized clinical trials (DCTs), for both investigators and patients. In October 2023, the US Food and Drug Administration (FDA) announced it was forming a Digital Health Advisory Committee to help the agency explore the complex scientific and technical issues related to digital health technologies (DHTs). This article looks at the challenges and opportunities presented by technology use in DCTs.
Technology is what makes decentralized clinical trials (DCTs) possible. Patients can sign electronic consent forms, wear sensors and devices to obtain crucial health data, and use telehealth for follow-up. And investigators can employ electronic clinical outcome assessments and other software platforms to gather key data remotely and maintain virtual contact with trial patients.
In a recent Citeline webinar, “Decentralized Trials: Where We’ve Been and Where We’re Headed,” panelists discussed the challenges and opportunities presented by technology use in DCTs.
Catherine Gregor, chief clinical trial officer, Florence Healthcare, says technology adoption can present its own set of tactical challenges, especially for those who may not be as tech-savvy as others.
“We may be used to technology in our lives, but that doesn’t mean that we know how to work with it, and not all technology is fit for purpose,” she says. “A lot of the tech that’s been in clinical trials has been designed from sponsors to sites, and so that creates a little bit of misalignment in workflow. Instead of complimenting clinical workflow, it tends to disrupt it.”
Gregor says the question is how to bridge that gap, as well as how to get past the point of too much technology.
“What is the right balance in terms of giving the sites resources that they need to conduct the trial and patients the resources that they need to participate, but not overwhelming them with, ‘I have a gadget for this type of read and I have a gadget for this type of read?’” she says.
According to Oracle, a single clinical study can often involve up to 30 different systems, each with its own credentials, interfaces, and validations. This can create a fragmented and frustrating experience for patients and make it difficult for sites to obtain a clear picture of that experience.1
Gregor recalls being in practice and having to manage multiple electronic consent tablets for every study she was running. “Which means my nurses who are managing nine or 10 studies have 10 tablets that they’re walking around with trying to consent patients,” she says. “That’s just ridiculous when you think about it.
"So, I think we as an industry have to talk about how we make that more seamless. Is it bring your own device? Do we always have to have one way or the highway, site’s own tech, sponsor’s own tech? Can we make them talk? Why does it have to be one version versus the other? Those have been my biggest challenges to date.”
It’s not only the devices that may not be compatible. Sites often have a variety of existing software platforms, while sponsors and CROs may have a completely different set. This can pose an issue when sites are selecting sponsors to work with. About 36% of sites responding to a Florence Healthcare survey say willingness to accept and integrate with existing site-based technology is the most important factor when choosing a sponsor.2
Gregor calls dealing with separate platforms “the swivel chair” method of data entry. “I’m literally taking from this system to put in this system so that everybody has access to it when that data flow should be seamless,” she says. “It’s seamless in other industries, and clinical research, it’s one of the few where we still have a person who has to move information from one system into another because we can’t figure out how to integrate that flow. And that’s on the vendors. The vendors need to solve that problem for their stakeholders, but we as stakeholders have to advocate for it.”
“Once we have that interoperability, once we’re able to make that data flow, there is the access question too,” says Lindsay Kehoe, senior project manager, Clinical Trials Transformation Initiative (CTTI). “Do the sites allow for the access that’s needed for research and for trials that are maybe coming out of the EHR [electronic health record]? There’s certainly some variation that we’re encountering across, and I think that there’s some work to be done from the site side, too.”
New FDA Advisory Panel to Focus on Digital Health Technologies
In October 2023, the US Food and Drug Administration (FDA) announced it was forming a Digital Health Advisory Committee to help the agency explore the complex scientific and technical issues related to digital health technologies (DHTs). These DHTs include artificial intelligence/machine learning (AI/ML), augmented reality, virtual reality, digital therapeutics, wearables, remote patient monitoring, and software.
The expertise and perspective provided by the committee will help improve the FDA’s understanding of the benefits, risks, and clinical outcomes associated with DHT use. The committee is expected to be fully operational in 2024.
This move follows the FDA’s December 2021 draft guidance on DHTs for remote data acquisition in clinical investigations. It is not clear whether this advisory panel will issue recommendations that will change the draft guidance, although it is possible.
In the release announcing the committee’s formation, Jeff Shuren, M.D., J.D., director of the FDA’s Center for Devices and Radiological Health, says as DHTs advance, the FDA must ensure it applies its regulatory authority in a way that both protects patient health and supports innovation.
“As one of our strategic priorities, our goal is to advance health equity in part through expanding access by bringing prevention, wellness, and healthcare to all people where they live — at home, at work, in big cities and rural communities,” Shuren says. “Digital health technologies are critical for achieving this transformation in care delivery.”
Source:US Food and Drug Administration (2023) FDA Establishes New Advisory Committee on Digital Health Technologies [Accessed on Oct. 20, 2023].
According to Gregor, talking about access means talking about the digital divide. She says she wonders if more technology-enabled trials are creating new barriers for patients.
“Not everybody has internet, not everybody has a mobile device. So how do we address those patients and not leave them behind?” she says. “And that gets to this question of optionality, which makes biostatisticians quiver in their boots because that means variability in data. So how do we move past that and what does that really look like? I don’t think anybody’s completely figured it out yet.”
On the question of whether the digital divide is going to create more inequality for patients, Jeff Thompson, senior director of strategic partnerships, GoodRx, says he would love to see what the data look like. “Which of those is the lesser of two evils? Which of those is less of a barrier of getting patients actually into trial?” Thompson asks.
“From GoodRx’s standpoint, we see a hundred million unique visitors a year to our ecosystem,” he adds. “We have 950,000 healthcare providers that use the platform. So being able to leverage that technology to broadly put the message about these trials out there in highly targeted places within the ecosystem I think is going to be a benefit to quickly filling these trials, getting the right patients in the trials, and ultimately getting therapeutics approved in a more timely fashion. Kind of a pivot, not really a challenge. I see it as a benefit of DCTs as we move forward.”
With DCTs, it is essential to maintain data integrity during integration, and to know when to say enough data has been collected.
“There are a lot of lessons learned with data,” Gregor says. “How do we get the data? How do we monitor the data? How do we make sure the data is handled in a way that is compliant with regulations and privacy?
“I think the lesson that I’ve seen is that most of the vendors have gotten very savvy at validating their systems and should be able to provide that validation upon review of ‘this is how we maintain privacy,’” she says.
In talking about personal security with data, Gregor says there needs to be trust that a different way of reviewing data will be acceptable. “Remote monitoring, risk-based monitoring, things that have been around for years that have been slow to adopt despite being enforced or endorsed by the FDA,” she says. “I think there is still a lot of challenge with that, and how do we take a population — clinical trials people are risk-averse by nature — and make them more open to letting there be a little more removed oversight of the data is the piece that I’ve seen a lot of people struggle with.”
“When we think about the oversight of the data, there’s probably a piece there that getting that patient input will maybe help move us in the right direction,” Kehoe says. “So, thinking a little bit about that patient centricity and considering what data will be shared back, what’s their willingness to use it and just transparency around how it’ll be captured, when, and how it’ll be shared back.”
“We’ve gotten really good at collecting data,” Gregor says. “We have all sorts of ways to collect data and we love data, and we get data hungry and we ask for all these endpoints. And sometimes it’s easy to forget that there’s a patient on the end of those endpoints.”
Gregor says it is important to dial down to the data truly relevant for approval, rather than focusing on data that’s nice to have but may not be necessary.
“I know ASCO [American Society of Clinical Oncology] talks a lot about reducing the amount of inclusion/exclusion criteria, reducing the number of endpoints in a trial to make sure that we’re focusing on what is truly valid to answer the question [rather than] what’s just, oh, but if we had this one more thing, we could answer this other question,” she says. “Because that one more thing, it means six more visits for a patient who doesn’t feel well or seven more blood pokes. How do we minimize that?
“I think that the decentralized element calls all of those up because when you remove barriers and you can have more access to data, there’s a tendency to want to grab. We have to move forward in a way that is thoughtful and diligent so that we don’t overburden on that side.”
Kehoe says it’s worth highlighting digital literacy and patient understanding and willingness to work with tech-enabled DCT approaches.
“When we think about best practices around data … certainly there’s likely the need … [for] feasibility and usability tests, focus groups gathering input early on from populations of interest in order to make certain that wearables, tech devices are going to be used appropriately,” she says. “And that not only are they going to have access to the technology, but also it’s good data that’s coming back and that there’s a level of integrity there.”
When it comes to adoptability around things like wearable tech, Gregor says there’s a certain comfort level that must be observed and accounted for.
“You’re going to have a tech-savvy level with patients, and then also making sure that the clinics that support those patients are educated on how to fix issues on those devices,” she says. “Because unfortunately, when something breaks on a patient, they’re not going to call Apple when they can’t get into their phone. They’re going to call the nurse and say, ‘This app doesn’t work and I’m not going to use it.’ So, your nurse or your coordinator has to know how to answer those questions. Creating technology that is not only easy to manage but has a great UI [user interface] and the staff are trained on how to support it, have been some of the biggest pieces around adoptability.”
Kehoe says another important point is being cognizant of how many endpoints the study is trying to collect. “Because if you’re asking a 15-year-old to contribute data over a course of 10 years, even if it’s remote, that’s going to be a tall order, retention will likely fall off,” she says. “I think we just need to be aware, whether it’s DCT or not, how we’re thinking about these trials overall.”
“If someone’s dropping off of a trial, maybe we ask them, why?” Thompson says. “Is it because it’s over 15 years? Is it because they’re not feeling well, they don’t want to drive to the study site? Or maybe it’s something that the regulatory folks or the sponsors of the study hadn’t even considered. But using technology to interact with those patients allows them to give that feedback, I think, in an easier way and allows it to be addressed more quickly, because it’s sort of top of funnel, top of mind.”
“I think asking the sites who deal with those patients too, and knowing if you want to know what’s going to make my patients tick, I’m going to … ask my nurses,” Gregor says. “Because they see them every day, and they can tell you, ‘This halter monitor is not going to work because my patients don’t like it, it itches, and they’re not going to wear it.’ So we know the data’s going to be crap. Why are we even putting it in the study?
“I think it’s really getting feedback from all the end users about what makes a difference and what is likely to be used. Because if it’s not used, it doesn’t matter how cool the device is or how much data it can gather if it’s sitting on a table and it’s not where it needs to be.”
The Association of Clinical Research Professionals (ACRP) surveyed members and stakeholders to determine how often they used specific decentralized clinical trial (DCT) technologies. Respondents reporting using operational technologies like electronic data capture (EDC), clinical trial management systems (CTMS), and e-Patient Reported Outcomes (ePRO) much more frequently than emerging technologies like telemedicine, wearables, and direct-to-patient (DTP) portals.
Figure 1: Frequency of Use of Select DCT Technologies
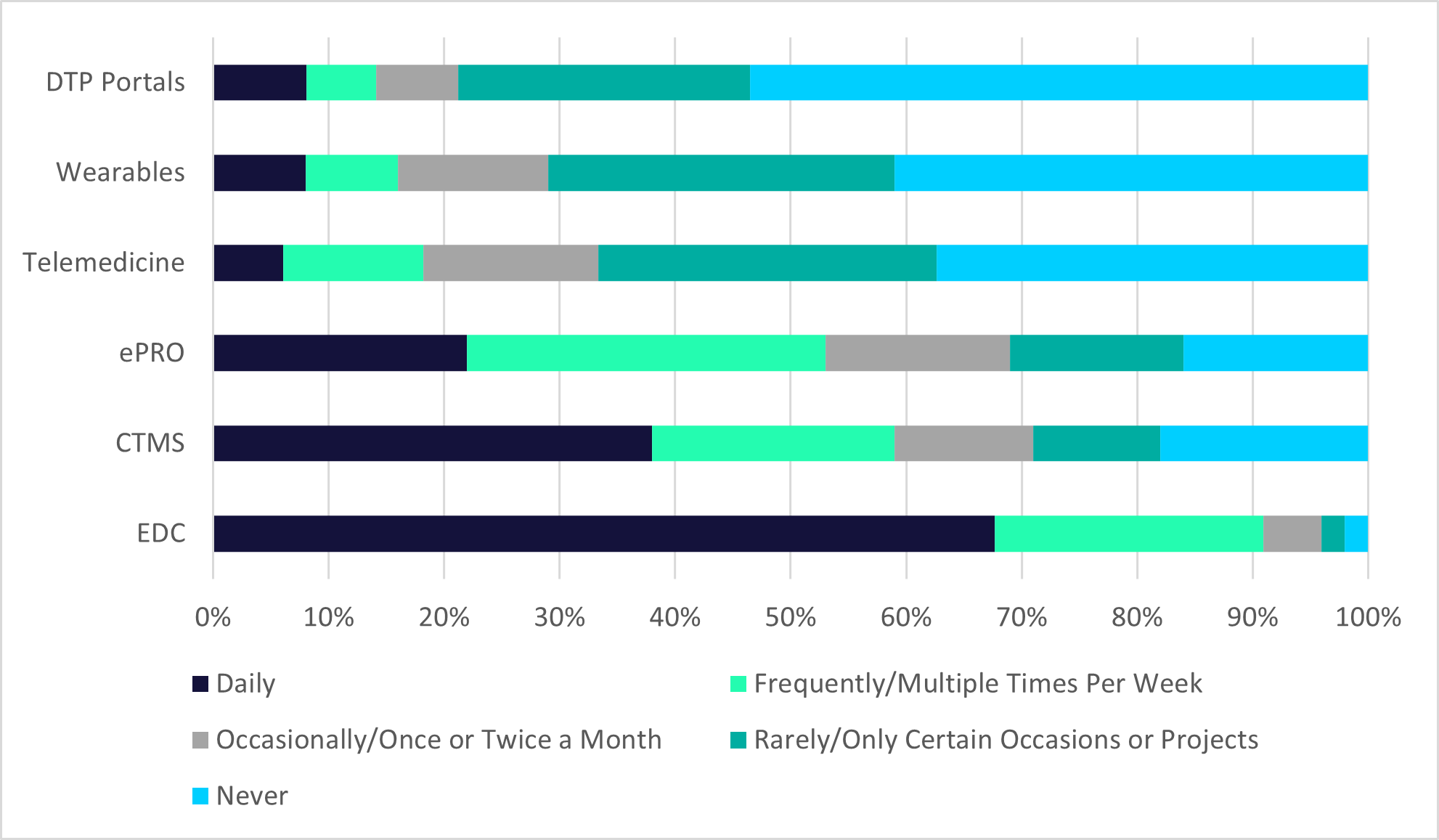
Source: The Association of Clinical Research Professionals (2022) Delivering on the Promise of Decentralized Trials: Unexpected Perspectives from Clinical Research Professionals. Available from https://acrpnet.org/delivering-on-the-promise-of-decentralized-trials-unexpected-perspectives-from-clinical-research-professionals/ [Accessed Oct. 19, 2023].
From a software perspective, Gregor says educating sponsors about trial modernization is going to be slightly different.
“From my end, we spend a lot of time with our sponsor customers and our CRO customers talking about what you can do in country with our tools and give guidance and then draw the lines and advise,” she says. “And sometimes we find that that guidance is taken. Sometimes there’s a slightly different interpretation. But we do have a lot of partner conversations about, ‘You can use for remote monitoring of regulatory documents, but not patient data in this country.’ Or, ‘You can have a qualified signature, but you don’t have a compliance signature,’ like these nuances. Our compliance team has spent a lot of time digging into that to understand all those little pieces and parts because it is quite complicated when you look at deploying a global study.”
“I think there’s a general interest from the sponsors to want to modernize,” Kehoe says. “You have the TransCelerates out there that have a whole kind of program devoted to modernizing clinical trials, and that is a conglomeration of pharmaceutical companies. [In] our industry in general, I think, this risk aversion is a big piece. And if we can keep communication between regulatory groups, pharmaceutical industries, making certain other stakeholders, data tech, patients are all at the table, it’s going to help move that needle a little bit forward.”
1 Oracle Health Sciences, Top 5 Challenges Of Decentralized Clinical Trials And How To Overcome Them. Available from https://www.oracle.com/a/ocom/docs/industries/life-sciences/ls-top-5-challenges-decentralized-clinical-trials-wp.pdf [Accessed Oct. 19, 2023]
2 Florence Healthcare (2023) 2023 State of Clinical Trial Site Enablement Technology Report. Available from https://florencehc.com/resource/2023-state-of-clinical-trial-site-enablement-technology-report/ [Accessed Oct. 16, 2023]




To enable the booking feature, please enable all cookies in your browser.