Citeline’s Patient Recruitment Pulse – March 2026 Edition
Blog
Patient Engagement & Recruitment


Welcome to the March 2026 edition of Citeline’s Patient Recruitment Pulse. In this monthly blog, we discuss the trends shaping today’s patient recruitment landscape. This month, we’re taking a closer look at some of the key findings of our white paper, Earning Their Trust: Challenges and Best Practices in Rare Disease Patient Recruitment.
Worldwide, 300 million people live with a rare disease, many of whom have inadequate treatment options or lack them altogether.
The good news is that research into rare diseases continues to grow. The 2025 edition of Citeline’s Annual Clinical Trials Roundup found Phase I–III clinical trial initiations for rare disease accounted for 30% of total trial activity in 2024.
Now, the not-so-good news: Finding patients for rare disease clinical trials can be an uphill battle, with obstacles including lack of awareness, delays in diagnosis, and trial design that doesn’t take the patient into account. Fortunately, there are ways sponsors can address the most common stumbling blocks in the recruitment process.
In our latest white paper, we outline some of the challenges and best practices in rare disease patient recruitment. Here are some of the key findings:
In a 2020 study by the National Organization for Rare Disorders (NORD), 88% of the 1,108 respondents living in the US with a rare disease said they would consider using an investigational treatment. But a 2025 Rare Patient Voice survey of 1,147 patients and caregivers in the US found that 57% were only “somewhat aware” of clinical trials.
This gap between willingness to participate and lack of information about clinical trials can be bridged by sponsors making information more accessible — particularly to a trusted resource like the patients’ healthcare providers (HCPs).
According to the 2020 NORD study, 28% of rare disease patient respondents said it took seven years or more for them to receive a correct diagnosis, and 38% of rare disease patient respondents reported receiving at least one misdiagnosis while on their diagnostic journey. A 2024 study conducted by NORD and the Rare Disease Diversity Coalition (RDDC) notes the increasing number of innovative rare disease therapies with narrow treatment windows. Any delay in diagnosis can keep rare disease patients out of clinical trials for such therapies, meaning they’ll never see any potential benefits.
 Jenifer Waldrop
Jenifer Waldrop
RDCC Executive Director Jenifer Waldrop says the 2024 study findings indicated “patients were not necessarily being asked to participate in clinical trials. Plus, patients were having challenges to participate in the trials themselves, given the construct and the way that the trials were designed.”
While the problem of delayed diagnosis is beyond the sponsor’s control, solutions are available that can really dig into data to help surface potential patients for rare disease trials. Citeline PatientMatch, for example, applies natural language processing (NLP) to both structured and unstructured clinical notes. This can enable detection of deeper clinical signals and identification of patients beyond standard coded diagnoses.
The problem of trial design not reflecting the day-to-day realities of rare disease patients isn’t new. For instance, when sponsors plan trials, they often fail to consider that the sites they choose may not be accessible to the patients they are trying to recruit, or that participants may need assistance with child or elder care.
Including patients and patient advocacy groups in the design stage of trial planning can help sponsors avoid these barriers. Once patients know they can assist with clinical trial design, they are often willing to participate. In a 2025 study by Rare Patient Voice, when asked if they would be interested in being involved in the design of a clinical trial in the future, 82% of respondents said yes.
Q: What would encourage you to participate in codesign activities?
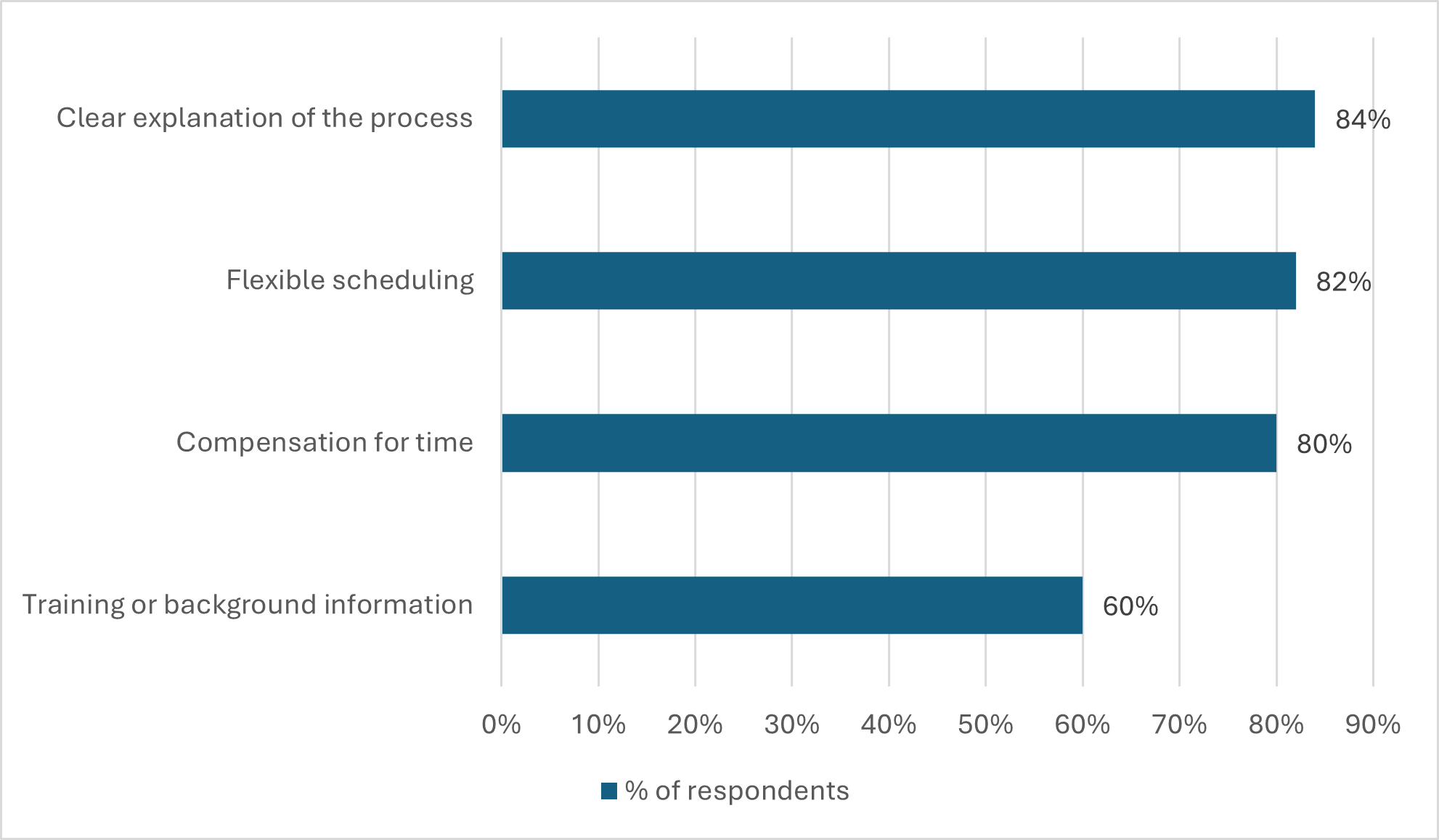
Source: Rare Patient Voice
 Harsha Rajasimha
Harsha Rajasimha
Lack of trust in pharma is another issue among rare disease patient communities. According to Harsha Rajasimha, Founder and Executive Chair of Indo US Organization for Rare Diseases, trust and transparency are critical to successful recruitment of rare disease patients.
Rajasimha recommends building relationships with rare disease communities and patient advocacy groups as much as one or two years before finalizing the trial protocol. “It’s important to engage, educate, and get educated by the patients and listening to them.”
Waldrop also emphasizes the importance of sponsors “showing that they’re there for the long haul. … Community partners can tell when engagement is episodic rather than sustained; multiyear commitments and ongoing education build credibility. If they continue to share how they really do care about the greater health of the community, then it provides a level of street cred and brand for them as a sponsor.”
For more details, download the white paper, which includes further insights on retention, decentralized trials, and the role of AI in rare disease trials.



To enable the booking feature, please enable all cookies in your browser.