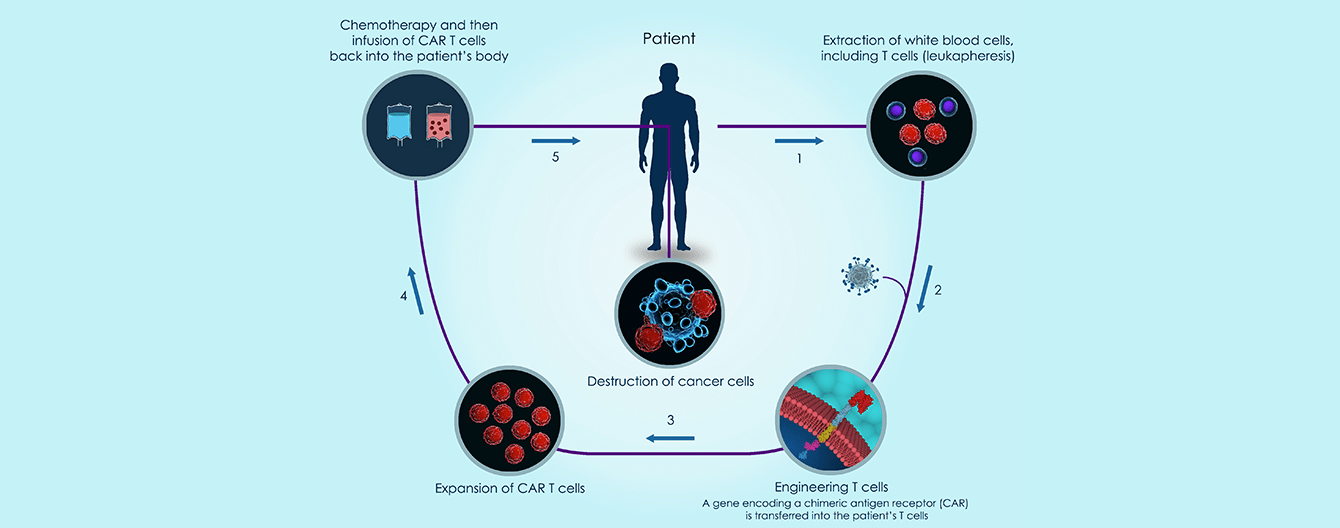
Chimeric antigen receptor T cell therapy (CAR-T cell therapy) has revolutionized the treatment of hematologic malignancies in patients with relapsed or refractory disease without other treatment options.
- CAR-T cell therapy is a groundbreaking treatment for certain cancers, but its full potential can only be realized if the industry ensures that clinical trials are diverse and inclusive.
- Despite the therapy’s potential, access remains highly restricted, particularly among minority and vulnerable populations.
CAR-T cell therapy and current demographics
- According to the Center for International Blood and Marrow Transplant Research (CIBMTR),179% of patients who received CAR-T cell therapy were Caucasian, 6% were African American, and 8% were Hispanic or Latino.
- Medicare data shows that 87% of patients receiving CAR-T cell therapy for B-cell lymphoma were Caucasian.
- An analysis2 of 4,396 patients revealed only 5.9% were African American.
Disparities in access and representation
- In seven pivotal CAR T-cell trials, only 2% to 5% of participants were African American.
- There is an extremely low participants/prevalence ratio, particularly in multiple myeloma, which is more common among African American persons. One explanation for this is that geographic distribution of clinical trials for CAR-T cell did not include 60% of the states with the highest proportion of African-American residents. This means new therapies are studied in populations that could differ dramatically from those to whom the therapies will be applied, these differences could have a major impact on real-world outcomes.
- A study of multiple myeloma patients found that African Americans were 49% less likely than whites to undergo stem cell transplantation. Even when controlling for variable access based on income and insurance, African Americans were still 37% less likely to undergo stem cell transplantation, suggesting that possible structural barriers exist within the healthcare system (e.g., referral bias, cultural barriers, conscious or subconscious bias among physicians), as well as possible decision-making differences between African American and white patients.
- Factors such as differences in social support and in the burden of comorbidities also limit access.
- CAR-T cell therapy is available in <4% of health care centers in the US, most of which are large academic medical centers. This results in additional travel and lodging costs, which are exacerbated by the fact that patients are required to have a caregiver and to remain in the vicinity of the institution in which they receive their therapy for at least one month after infusion. Thus, there is also the short-term loss of income for both the patient and their caregiver that must be factored into the overall cost of treatment. Taken together, the real-world cost of CAR-T cell therapy can range from $700,000 to $1 million, which may make the treatment unaffordable for those patients without robust financial and/or social support. The degree to which this cost is covered is currently unclear and dependent on type of insurance coverage, especially private vs. public coverage.
Fostering equitable access and inclusivity
- Availability can be increased by involving and preparing community hospitals and other healthcare facilities that can deliver CAR-T cell therapy safely should be an immediate priority to improve access.
- To guarantee universal availability, a change in culture is required, by both the scientific community and governments, which must develop health legislation and health economics focusing on diversity, equity, and inclusion (DEI).
- Organizations should consider each community's demographic, its culture, history of racism, and local events that might influence whether people are hesitant to seek care when needed.
- National programs and drug companies must support patients with a financial infrastructure designed to offset costs and diminish logistical burdens in providing care that leads to equitable outcomes regardless of socioeconomic status and neighborhood opportunity.
- Improving CAR-T cell access from a DEI perspective involves a multifaceted approach implementing strategies and initiatives that address the barriers and disparities faced by diverse populations. Product-related efforts, such as escalating manufacturing and increasing cell therapy delivery sites, will likely provide the most immediate improvements. However, this will only provide a partial fix to a much deeper problem.
Addressing socioeconomic disparities requires a multi-pronged approach involving policy interventions, patient support programs, education, and increased equity in clinical trials. This requires implementing policies and initiatives that prioritize equitable healthcare delivery, reduce financial barriers, and enhance representation in clinical trials. Furthermore, collaboration among healthcare providers, policymakers, and community organizations can help identify and overcome barriers to access, promote culturally competent care, and develop targeted outreach programs.
1 https://cibmtr.org/Files/Meetings/Working-Committee-Meeting-Materials/Cellular-Immunotherapy-for-Cancer-Meeting-Materials/2022-Cellular-Immunotherapy-for-Cancer-Agenda.pdf
2 https://www.astctjournal.org/article/S2666-6367(22)01224-6/pdf