CGT Development Is Booming, but Manufacturing Workforce Must Keep Up
Article
Commercial

ANALYSIS
By Amanda Micklus and Shardha Millington
Research into workforce demands in cell and gene therapy, conducted by Citeline in collaboration with the Alliance for Regenerative Medicine (ARM) and performed with industry and academic subject matter experts, was prompted by an executive order issued by the Biden administration in September 2022 to launch the National Biotechnology and Biomanufacturing Initiative. The order aims to coordinate the US government’s approach to advance biotechnology and biomanufacturing toward innovative solutions in multiple sectors. ARM shared the results of the qualitative and quantitative research publicly in its workforce gap analysis report, published in March 2023.
The cell and gene therapy sector has expanded tremendously, with the number of drugs in development growing nearly six-fold from 511 development-stage candidates in 2013 to just over 3,000 in 2023, according to Citeline’s Pharmaprojects. Attempting to meet the high demand for specialized skill sets to produce these advanced therapies has been challenging, with the workforce traditionally struggling to keep up and fill certain roles. In recent years, though, the supply of skilled workers has somewhat improved, albeit for negative reasons: Increasing layoffs and workforce reductions in the overall biotech industry have caused a boost in the number of people looking for new jobs.
Even so, industry and academic stakeholders representing the cell and gene therapy sector who participated in the research believed that this increase in supply was only temporary, and that demand will pick up again for skilled workers, especially across several key areas including manufacturing. As opposed to traditional small molecules or biologics, manufacturing in advanced cell therapies requires highly specialized and unique skill sets and considerations, including novel approaches to upstream processing, downstream processing, analytical development, and logistics.
Specifically, one of the biggest gaps is in entry-level manufacturing, where there is a need for training facilities in good manufacturing practices (GMP). Experts believe an effective way to solve this would be to establish GMP training facilities to widen the applicant pool in entry-level manufacturing roles. To boost the workforce further, wider availability of training could increase the number of qualified candidates in other functions, too, including quality and development.
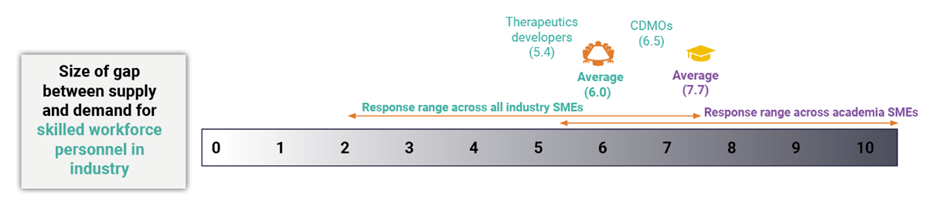
Ultimately, the research showed the gap between demand and supply still very much exists and is substantial (see Exhibit 1). To quantify the gap, industry and academic subject matter experts in cell and gene therapy were asked to rate the size of the gap in the workforce on a scale of 0 to 10, where 0 meant no gap and 10 meant an extremely big gap. According to academic stakeholders, the extent of that gap is large, with academic participants scoring an average 7.7 out of 10. Industry participants were slightly more optimistic, but still weighted toward a large gap in supply and demand with an average score of 6 out of 10. Among the industry representatives, therapeutics developers were even more positive, likely because of the current widened pool of potential applicants due to industry downsizing, giving an average score of just 5.4 out of 10.
Source for all exhibits: Citeline | Alliance for Regenerative Medicine primary research
Collaboration Between Academia and Industry
Academic and industry stakeholders have identified that in-person training is generally preferred due to the hands-on nature of the education and the benefit of being able to tailor training to the institution or company in question, such as training on very specific manufacturing processes and GMP requirements. While methods such as virtual reality (VR) have been explored, usage is still limited with only 33% of interviewed industry stakeholders mentioning they had experience with VR training. Of those industry stakeholders who have used external training resources, the ones reported to be most useful are National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL), and Master of Science (MSc) programs such as those provided by Case Western Reserve University, Johns Hopkins University, University of Southern California, and Wake Forest University.
Although internal company training is seen as highly important and beneficial, alignment and collaboration between academia and industry is where the greatest success can often be found for skill development: 60% of academic respondents have partnerships with industry for workforce training and report a success rate of 78%. The advantages are numerous; the greatest benefit is the job opportunities for students who can use the industry partner as a doorway into employment. Other advantages to partnerships include the guidance and funding that industry can provide for workforce training development based on real industry needs.
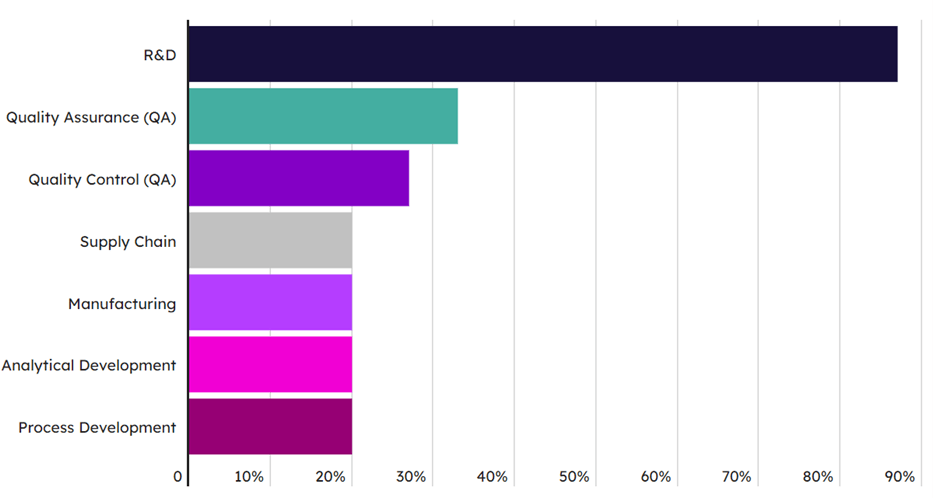
Importantly, in addition to an understanding of the current training landscape, feedback from the academic and industry stakeholders in this research has provided good direction for where further development on education and training can be focused to close the gap between supply and demand for skilled industry workforces. An example includes the need for further training on more specific niches within regenerative medicine subjects and workstream areas. Training is currently heavily focused on the general topic of “research and development,” with the more niche elements such as supply chain, analytical development, and process development receiving less coverage (see Exhibit 2). Similarly, the coverage of regenerative medicine areas in academic programs focuses highly on genetically modified cell therapies, gene therapies, and non-genetically modified cell therapies, yet often neglects topics such as gene editing and tissue engineering. In response to this deficit in the curriculum, academic institutions are looking to both expand their offerings to cover a broader range of subject material, but also offer courses that are more specific and focus-in on an individual workforce function, e.g., offering courses that focus solely on supply chain.
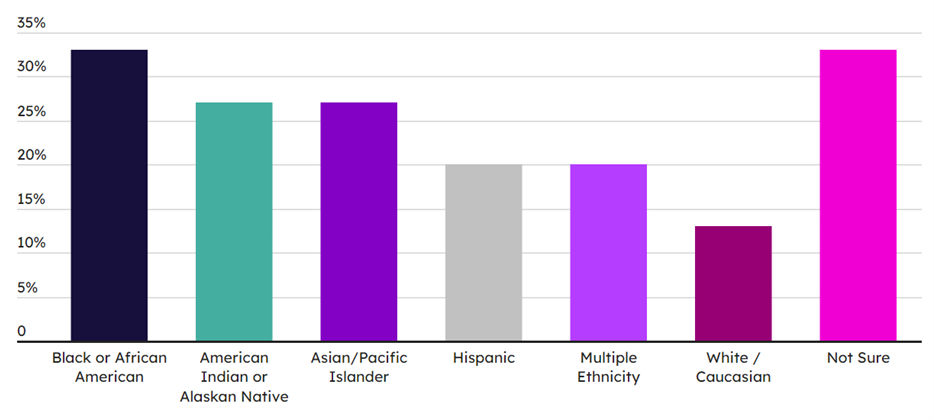
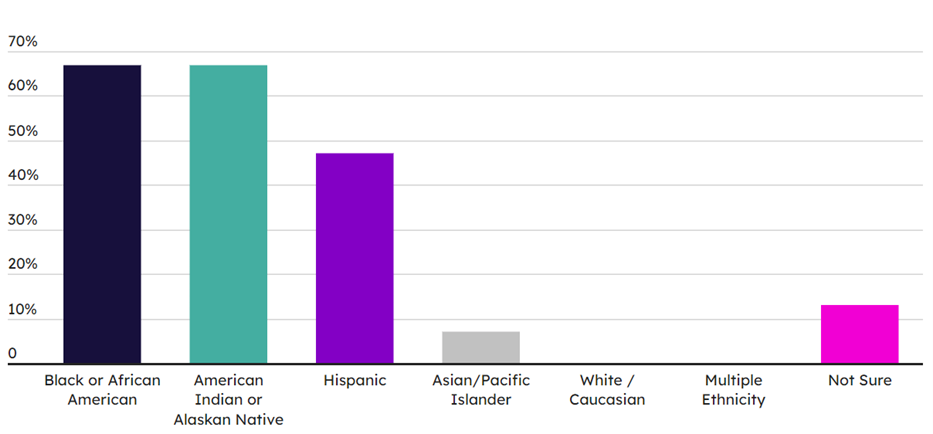
Although improvement opportunities have been identified and offerings are looking to be expanded, there are long-standing barriers to the access and availability of training that need addressing. Among the more commonly mentioned challenges is a lack of awareness and interest. Besides the contribution of the “bad pharma” image, the low visibility of training programs is associated predominantly with the difficulty that disadvantaged and under-represented individuals have in envisaging themselves in industry roles as a result of poor exposure to opportunities. Cost barriers also exist, not just in terms of training cost but also additional personal expenses such as childcare while a parent is in class, accommodation, and travel. These access issues for certain sub-populations and minorities are reflected both in the figures seen for difficulty of enrollment in workforce training, where only 13% of respondents felt enrollment was “not at all difficult,” but also in the demographics of training programs and the applicant pool for workforce (see Exhibits 3 and 4).
To combat these barriers, some solutions have been proposed including the sponsorship of workforce training by companies, leveraging networks such as NIIMBL for partnerships with community colleges, and training schoolteachers to better position them to educate the new generations on the workforce opportunities that are available.
Emerging Biotech Hot Spots Offer Untouched Resource Pool
The demand for skilled workforce stretches across the development and production chain for cell and gene therapies and will continue to heighten and apply pressure on CDMOs and therapeutics developers as the number of programs progressing through the pipeline increases. This is particularly the case for CDMOs that intensify recruiting in response to client activities, i.e., in line with approvals and need for manufacturing-scale-up. The biggest threats employers face regarding recruiting efforts to meet their demand are a lack of established training programs, compensation competition, and job hopping. The inability for therapeutics developers to compete with the high compensation and salary competition from CDMOs is also a big challenge when it comes to retention of current employees, according to interviewed industry stakeholders. This is something felt by academic employers, too, who in turn struggle to compete with the salaries that industry as a whole can afford to offer. Academic experts also highlight that industry needs to branch out beyond traditional forms of recruitment to have greater recognition of, for example, the value of shorter (two-year) degrees and qualifications. Workers graduating from these shorter-term programs could expand the eligible workforce in the cell and gene therapy sector, especially in manufacturing.
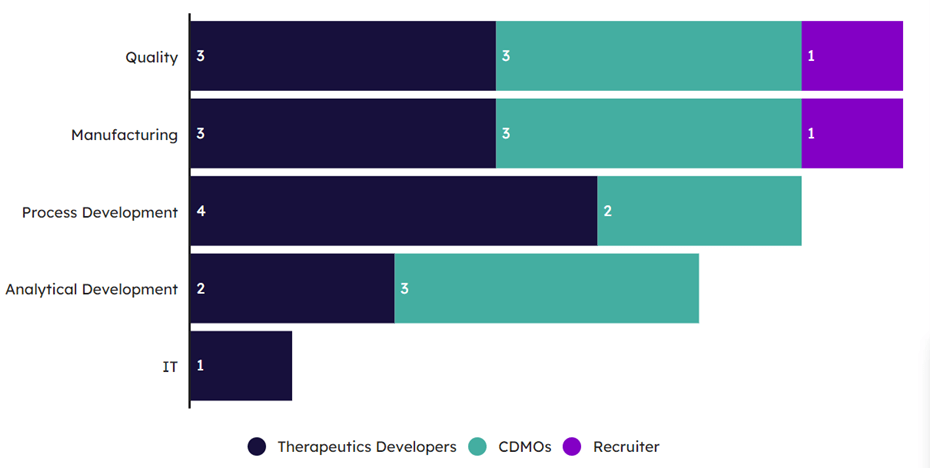
Although the aforementioned recruitment challenges apply across the board, the workforce functions in regenerative medicine that see the greatest challenges are quality, manufacturing, and process development (see Exhibit 5). These functions experience the longest recruitment times due to a combination of the need for highly technical skills and the lack of a skilled workforce that can fill those roles. The consequences of the difficulty faced in finding sufficient talent is only considered to have negatively impacted clinical development timelines by a minority of 40% of respondents; however, when timelines have been altered, the impact is usually moderate to high.
An interesting idea to remedy the lack of skilled workforce available is to recruit outside the regenerative medicine, or even the scientific, sector and employ individuals from alternative backgrounds such as ex-military leadership roles, UPS or FedEx workers in shipping/supply chain positions, and lab technicians experienced with blood samples. These individuals will all possess transferable skills in communications, logistics, structure, safety, and project management which can readily be applied in a cell and gene therapy context.
Looking to specific geographic areas can help with recruitment, based on how concentrated the regenerative medicine workforce is across the US. Hot spots of talent have formed around the sites of cell and gene therapy developers, CDMOs, and universities/academic centers (see Exhibit 6). Taking advantage of these geographic areas can help employers easily identify local talent; however, it also exacerbates the issue of job hopping due to the high numbers of competitors within the same vicinity. Although these pockets exist, we are now seeing new areas emerging as biotech clusters, which can offer an untouched pool of talent, including: Denver and Boulder (Colorado); Cleveland, where the state is providing tax incentives or lump sums to encourage relocation; Rockville (Maryland), where secondary education is focused on biotech; Philadelphia, which has a low cost of living relative to other hot spots; and Houston, which is building large lab, office, manufacturing, and training spaces.
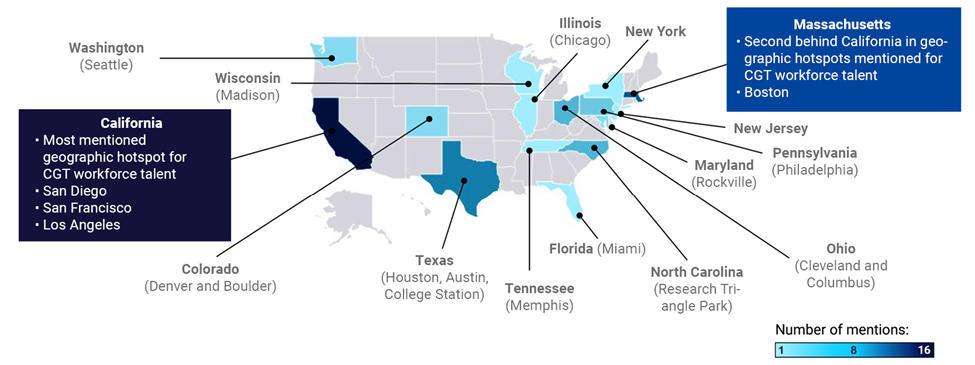
Note: Darkness of colors on map represent number of mentions by industry SMEs; the darker the color, the more mentions
Source for all exhibits: Citeline | Alliance for Regenerative Medicine primary research
Conclusions
Industry and academic stakeholders in the cell and gene therapy sector have noted many challenges in the supply and demand of hiring personnel in manufacturing and other functional areas, and have identified training gaps and recruitment issues. Despite these challenges, industry and academic SMEs are optimistic about the future, proposing solutions such as building more training suites and facilities, especially for entry- or low-level manufacturing; subsidizing expenses of training consumables; and focusing training on specific functional areas where there is a high unmet need, such as process and analytical development. Additionally, to ensure there is a strong future generation in the workforce made up of a diverse pool of candidates, key stakeholders can come together to provide educational initiatives, starting even at the elementary level, to increase awareness of regenerative medicine career paths. There are many opportunities for cell and gene therapy leaders to build a strong base as the industry prepares for more advanced therapies to enter the market.

To enable the booking feature, please enable all cookies in your browser.